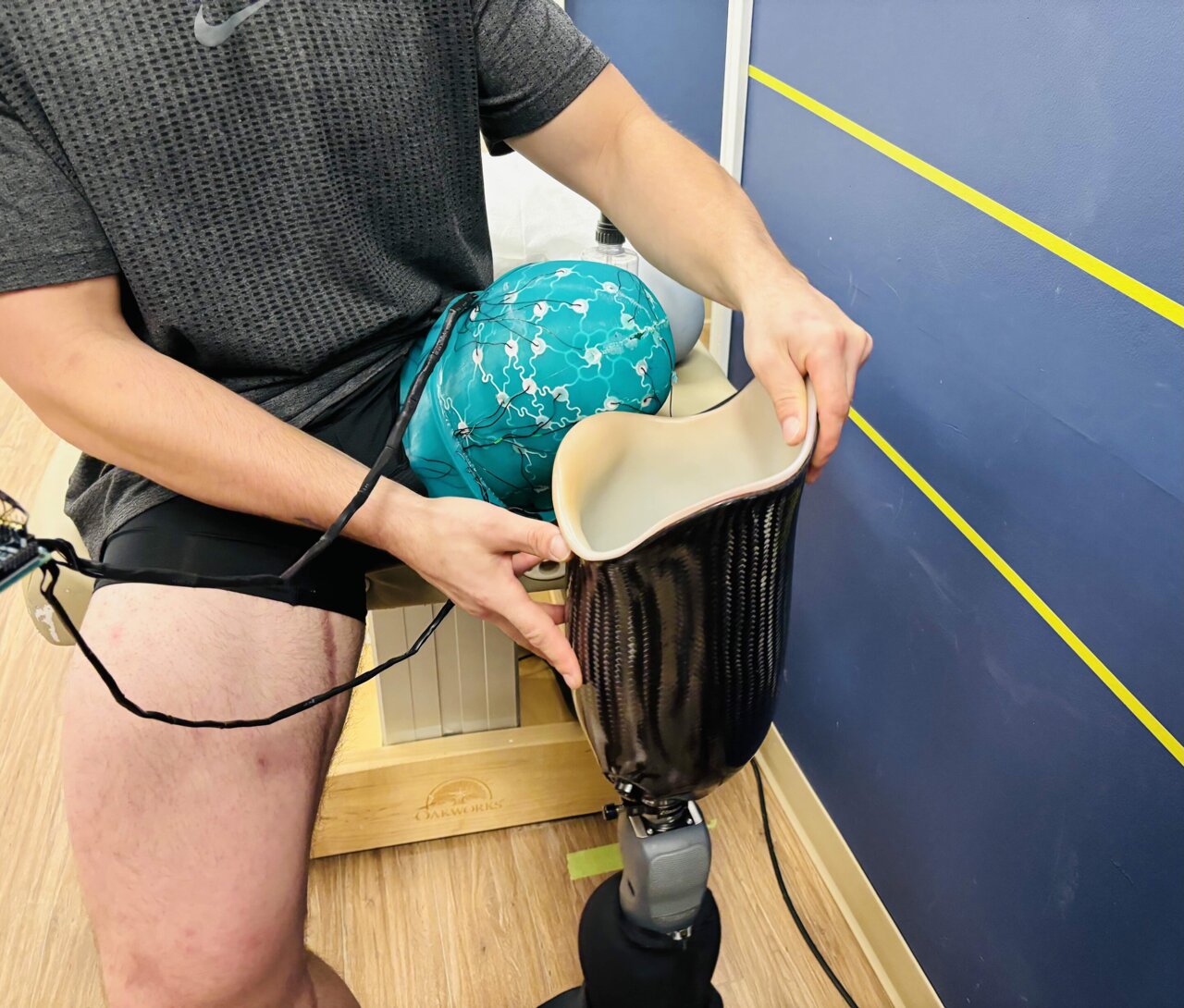
Phantom Neuro has received approval to begin its first-in-human Early Feasibility Study of Phantom X, marking a significant step forward for a company positioning itself at the intersection of prosthetics, neuromuscular interfaces, and robotic control. The study, known as CYBORG, will be conducted in Australia and is designed to evaluate the safety, efficacy, and usability of the company’s implantable muscle-machine interface in people with upper-limb loss.
According to the company, Phantom X is intended to help individuals with limb loss control prosthetic hands and other robotic devices through more natural, intuitive movement. Rather than relying on brain surgery or direct nerve intervention, the system uses a small implantable sensor array placed under the skin in the residual limb to interpret neuromuscular activity generated by muscles and translate those signals into device control. Phantom Neuro says this approach is designed to avoid some of the risks associated with more invasive neural interface technologies while still aiming to deliver more seamless prosthetic control.
The approved Early Feasibility Study will enroll up to 10 participants with unilateral transradial, or below-elbow, upper-limb amputation. The trial will take place in Melbourne, Australia, with surgical procedures performed at Cabrini Health. Prosthetic care and rehabilitation support for participants will be provided by ProMotion Prosthetics and Enable Rehab.
Phantom Neuro says participants will undergo a single outpatient implantation procedure, followed by around two weeks of recovery before they are fitted with a Phantom X-compatible prosthetic device. Each participant will then be followed over an approximately 20-week study period. The company has stated that data from the Australian study is expected to support a future U.S. FDA Investigational Device Exemption application and, later, a pivotal trial.
The company had already built regulatory momentum before this latest milestone. In March 2025, Phantom Neuro announced that Phantom X had received both FDA Breakthrough Device designation and entry into the Targeted Acceleration Pathway programme, which are intended to support the development and review of promising medical technologies. This new first-in-human approval therefore represents a move from preclinical and regulatory positioning into actual human implantation and real-world testing.
For the upper-limb prosthetics sector, that transition matters. One of the biggest ongoing challenges in advanced prosthetic care is translating user intent into reliable, intuitive movement without placing excessive cognitive burden on the user. Phantom Neuro’s proposition is that a minimally invasive muscle interface could narrow the gap between what the user wants to do and what the prosthesis actually does. That claim still needs to be validated in humans, but the new study is the first real opportunity to test it clinically. This assessment is an editorial inference based on the company’s stated goals and the trial design.
From an IMEA CPO perspective, the development is worth watching closely. While the CYBORG study is being conducted in Australia, innovations that improve intuitive prosthetic control have clear long-term relevance for rehabilitation systems globally, including emerging O&P markets across IMEA. If Phantom X or similar platforms can demonstrate clinically meaningful control improvements with a less invasive implantation pathway, they could influence future expectations around advanced upper-limb prosthetic fitting, rehabilitation protocols, and multidisciplinary care models. This is a forward-looking interpretation rather than a confirmed commercial outcome.
At this stage, however, the story is still an early clinical one. The study is small, focused on initial safety and usability, and limited to a defined amputee population. Even so, first-in-human approval is a meaningful milestone for any company developing a next-generation human-machine interface, and Phantom Neuro has now crossed that threshold.